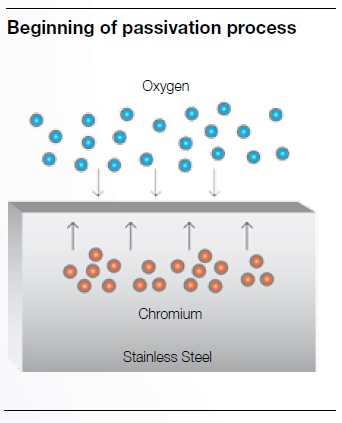
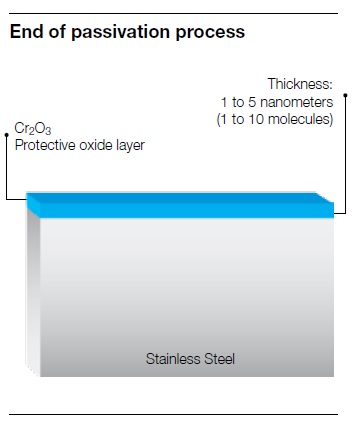
Stainless steel is generally able to resist corrosion thanks to the formation of a passive, chromium-oxide layer that forms on its surface, called passivation.
Passivation occurs whenever the chromium contained in stainless steel comes in contact with oxygen from ambient air. The chemical reaction will form a passive chromium oxide layer, which will protect the stainless steel surface. In order to maximize thickness and uniformity of the chromium oxide.